|
There is a maximum of two electrons that can occupy the s orbital. The principal quantum number n fills the s orbital. They are generally harder and denser than alkali metals, have 2 electrons in their outermost s sub-shell, and each make a distinct color in their flames. The s-block in the periodic table of elements occupies the alkali metals and alkaline earth metals, also known as groups 1 and 2. Soluble barium compounds are very toxic, while radium in all forms is very toxic, due to its radioactivity. The alkaline earth metals are found in column 2 on the left side of the Periodic Table. The list of elements that are alkaline earth metals is short. Magnesium and calcium are necessary for organisms, while strontium has been found to be beneficial to bone growth in controlled conditions. The alkaline earths are the elements located in Group IIA of the periodic table. 
Soluble beryllium and barium compounds are very toxic. Barium and radium sulfate/carbonate are among the most insoluble compounds known.Īll alkali earth metals are silvery-gray base metals. Strontium and barium burn in air to form oxide and nitride. Beryllium and magnesium form a thin protective oxide layer which limits oxidation, though magnesium's layer does not resist water. It is a dense gray metal, which has a pale greenish glow in the dark.Īlkali-earth metals, except for beryllium, react slowly with water. Radium, atomic number 88, is the only radioactive member of the group. It is a silvery-gray metal, which burns with a green flame. valance : All the alkali metals have an electron in their outermost shell and all the alkaline earth metals have two outer electrons. Strontium, atomic number 38, is a reactive metal which burns with a reddish or crimson-red flame.īarium, atomic number 56, is the heaviest stable member of the group. Electron configuration: Alkali metals have the electronic configuration of Noble gas ns1 and alkaline earth metals have, Noble gas ns2 electronic configuration. It rapidly tarnishes in air and burns with an orange flame. Magnesium burns with an extremely bright white flame, famous for its strength.Ĭalcium, atomic number 20, is the lightest alkaline earth metal, with a density of 1.55 g/cm 3, lighter than both beryllium and magnesium. It forms a protective oxide coating which slows its corrosion. Magnesium, atomic number 12, is the second lightest alkaline earth metal (1.738 g/cm 3). Beryllium's properties are closer to that of aluminium, a property referred to as diagonal relationship. Despite its position, it is not the lightest alkaline earth metal density-wise (1.85 g/cm 3). 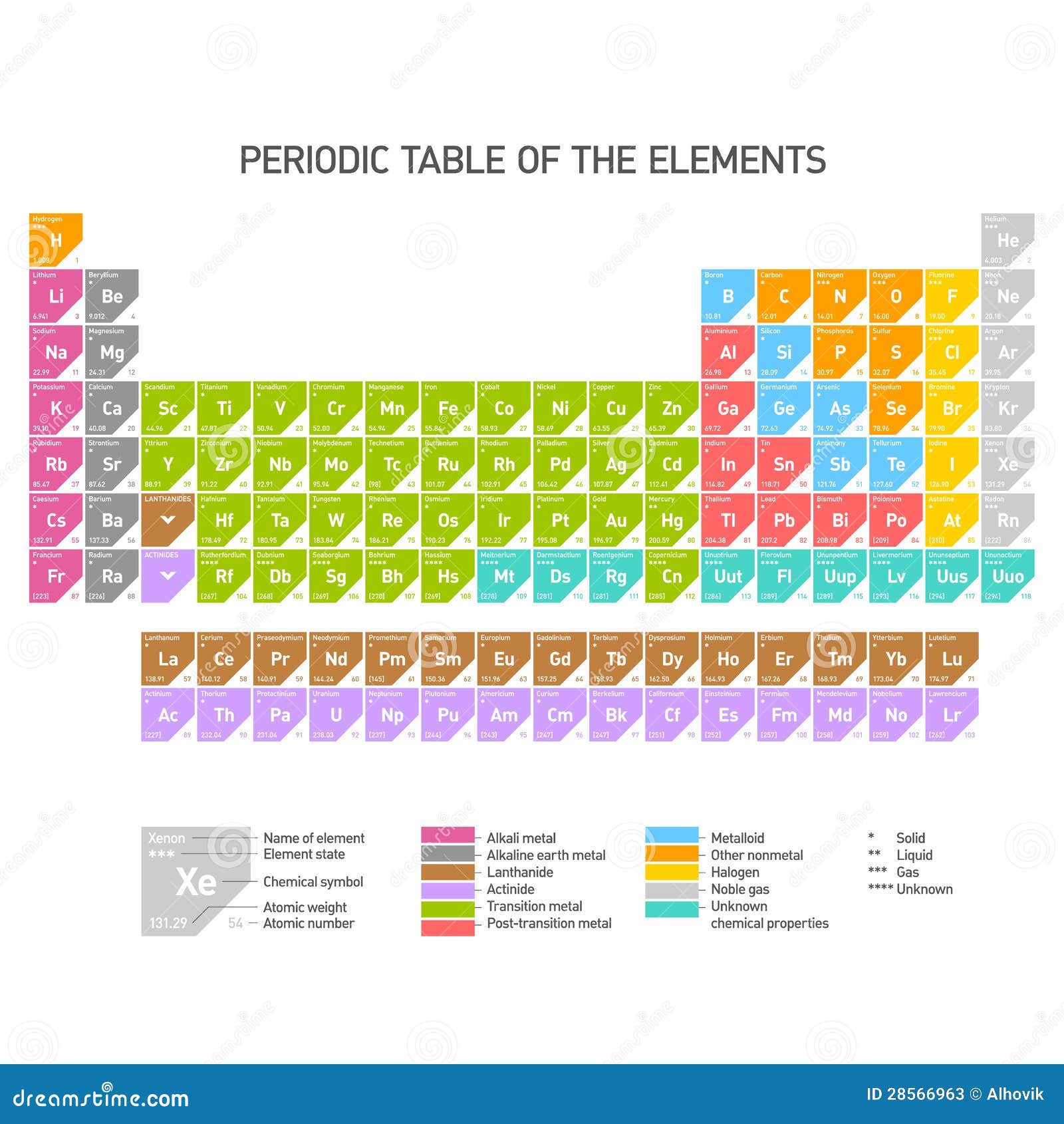
1 Elements in the alkaline earth metal groupsĮlements in the alkaline earth metal groups Berylliumīeryllium, atomic number 4, has the highest melting point of the alkali metals.The halides are ionic crystals, except for beryllium chloride, which is a covalent compound. 
All of the alkaline earths react with halogens to form halides.All the alkaline earths, except for beryllium, form corrosive alkaline hydroxides.These elements are shiny and silver-white as pure metals, although they usually appear dull because they react with air to form surface oxide layers.They are common in a wide variety of compounds and minerals. However, all of these elements do occur naturally. Because of their high reactivity, the alkaline earths are not found free in nature. potassium calcium lead copper Explanation: Alkaline earth metals are present in a group 2 in the modern periodic table. The alkaline earth metals are very reactive, although less so than the alkali metals. The alkaline earth metals or simply alkaline earths are recognized as an important group and family of elements.The elements readily form divalent cations (such as Mg 2+and Ca 2+).Relatively low melting points and boiling points, as far as metals are concerned.Two electrons in the outer shell and a full outer electron s shell.Summary of Common Alkaline Earth Properties
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
